❄ Freezing Point of Water at Altitude Calculator
Estimate the tiny freezing-point shift from elevation, pressure, and dissolved solids.
| Altitude | Pressure | Shift | Freeze |
|---|---|---|---|
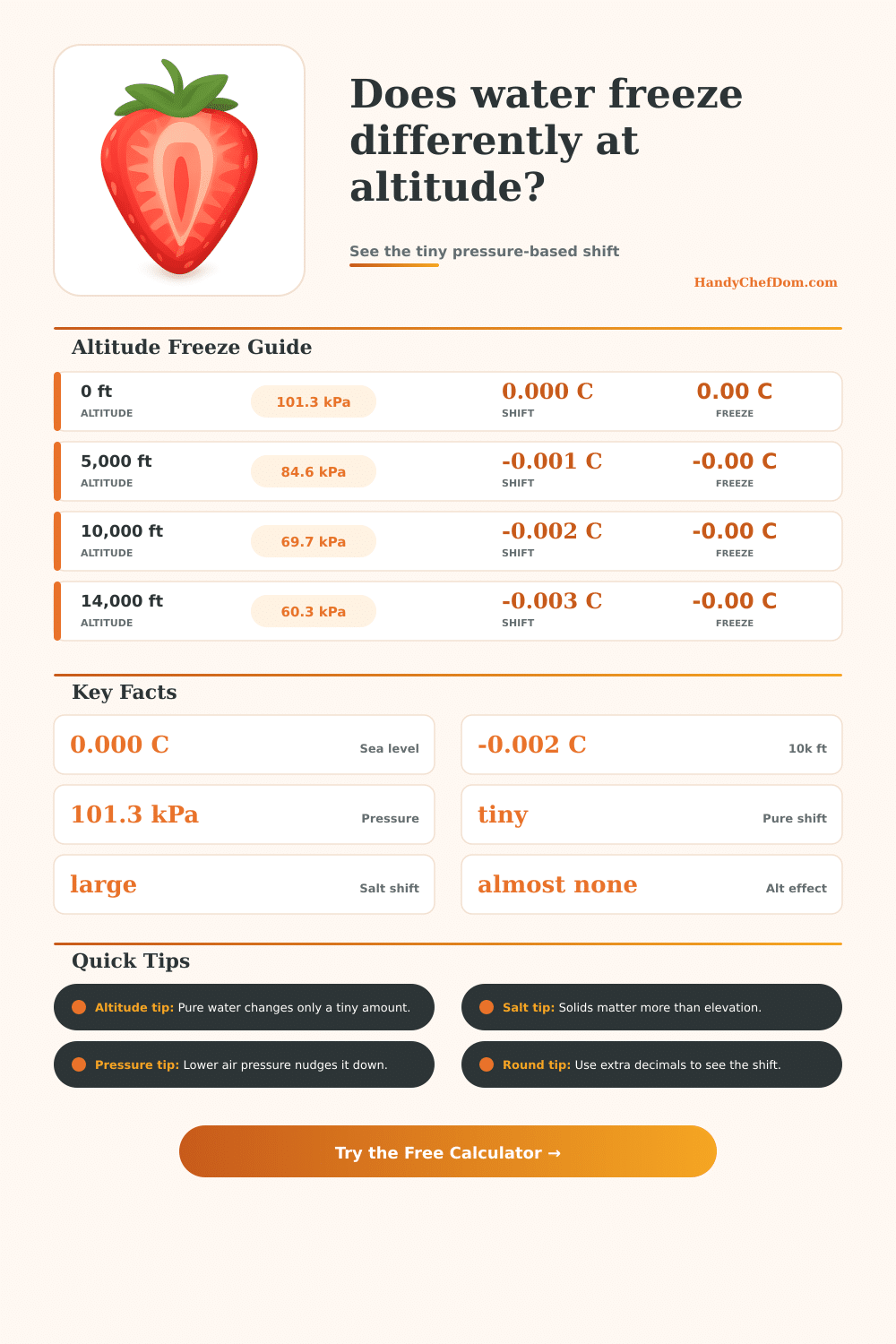
| 0 ft | 101.3 kPa | 0.000 C | 0.00 C |
| 5,000 ft | 84.6 kPa | -0.001 C | -0.00 C |
| 10,000 ft | 69.7 kPa | -0.002 C | -0.00 C |
| 14,000 ft | 60.3 kPa | -0.003 C | -0.00 C |
| Type | Shift | Use | Note |
|---|---|---|---|
| Distilled | 0.000 | pure | clean |
| Tap | 0.001 | house | tiny |
| Mineral | 0.003 | mineral | small |
| Brine | 1.900 | salted | strong |
| Place | Ft | KPa | Note |
|---|---|---|---|
| Sea level | 0 | 101.3 | base |
| Denver | 5280 | 83.4 | dry |
| Flagstaff | 6900 | 77.5 | high |
| Summit road | 12000 | 65.7 | thin |
| Effect | Pure Water | Salt Water | Takeaway |
|---|---|---|---|
| Altitude | tiny | tiny | near zero |
| Solids | tiny | strong | main driver |
| Pressure | tiny | tiny | very small |
| Roundoff | 0.00 | 1.90 | keep decimals |
| Altitude | Pressure | Shift | Freeze |
|---|---|---|---|
| 0 ft | 101.3 kPa | 0.000 C | 0.00 C |
| 5,000 ft | 84.6 kPa | -0.001 C | -0.00 C |
| 10,000 ft | 69.7 kPa | -0.002 C | -0.00 C |
| 14,000 ft | 60.3 kPa | -0.003 C | -0.00 C |
| Type | Shift | Use | Note |
|---|---|---|---|
| Distilled | 0.000 | pure | clean |
| Tap | 0.001 | house | tiny |
| Mineral | 0.003 | mineral | small |
| Brine | 1.900 | salted | strong |
| Place | Ft | KPa | Note |
|---|---|---|---|
| Sea level | 0 | 101.3 | base |
| Denver | 5280 | 83.4 | dry |
| Flagstaff | 6900 | 77.5 | high |
| Summit road | 12000 | 65.7 | thin |
| Effect | Pure Water | Salt Water | Takeaway |
|---|---|---|---|
| Altitude | tiny | tiny | near zero |
| Solids | tiny | strong | main driver |
| Pressure | tiny | tiny | very small |
| Roundoff | 0.00 | 1.90 | keep decimals |
| Altitude | Pressure | Shift | Freeze |
|---|---|---|---|
| 0 ft | 101.3 kPa | 0.000 C | 0.00 C |
| 5,000 ft | 84.6 kPa | -0.001 C | -0.00 C |
| 10,000 ft | 69.7 kPa | -0.002 C | -0.00 C |
| 14,000 ft | 60.3 kPa | -0.003 C | -0.00 C |
| Type | Shift | Use | Note |
|---|---|---|---|
| Distilled | 0.000 | pure | clean |
| Tap | 0.001 | house | tiny |
| Mineral | 0.003 | mineral | small |
| Brine | 1.900 | salted | strong |
| Place | Ft | KPa | Note |
|---|---|---|---|
| Sea level | 0 | 101.3 | base |
| Denver | 5280 | 83.4 | dry |
| Flagstaff | 6900 | 77.5 | high |
| Summit road | 12000 | 65.7 | thin |
| Effect | Pure Water | Salt Water | Takeaway |
|---|---|---|---|
| Altitude | tiny | tiny | near zero |
| Solids | tiny | strong | main driver |
| Pressure | tiny | tiny | very small |
| Roundoff | 0.00 | 1.90 | keep decimals |
The freezing point of waters changes when the altitude change, and the freezing point of water changes because the air pressure change. At sea level, the freezing point of water are 0 degree C because the air pressure is at its highest. As the altitude change to higher altitude, the air pressure decrease, which leads to a decrease in the freezing point of water.
The freezing point of water drop at a very small rate with changes in altitude; the freezing point of water only drop by thousandths of a degree as the altitude increase. For instance, at 5,280 feet in Denver, the freezing point of water drop by 0.002 degree C.
The relationship between air pressure and altitude are not a straight line, so air pressure does not decrease at a constant rate as the altitude increase. Instead, the atmosphere follow a predictable curve known as the barometric formula.
How Altitude and Salt Change Water’s Freezing Point
This barometric formula model the decrease in air pressure with altitude. While it may seem as if air pressure drop at a linear rate as the altitude increases, the drop in air pressure with altitude is, in fact, non-linear. The calculator that model the relationship between air pressure and altitude will provide an appropriate kilopascals for the given height, even without manually performing the equations for that calculation.
However, you should utilize the manual mode of the calculator to model the relationship between air pressure, altitude, and freezing point of water, especially when the weather are changing and air pressure is changing along with the weather pattern. The type of water is another important factor in the freezing point of water, and this factor is more important than the change in altitude. Pure water and distilled water will have the same freezing point regardless of the change in altitude, due to the lack of dissolved solid in these types of water.
Tap water contain minerals that will lower the freezing point of the water. Salty water will lower the freezing point of the water the most due to the increase in the amount of dissolved solid in the water. While the altitude can change the freezing point of water by thousandths of a degree, the addition of salt can change the freezing point of water by several whole degree.
Thus, the amount of dissolved solid in the water is one of the major factor in determining the freezing point of that water. You can see these factor at work in various scenario. For instance, snow that melt at 10,000 feet will have a freezing point that is 0.002 degree lower than water at sea level.
However, if the mountaineer add an electrolyte mix to the melted snow water, the freezing point will drop by more than 0.15 degree. Thus, the electrolyte is the more important factor in this scenario. Another example would be farmer who use brackish water from there well.
The salt in the water can change the freezing point of the water in the reservoirs by nearly two degree. Thus, the salt level in the water are the more important factor for these farmer than the altitude of their farm. Precision is an important factor in freezing point measurement.
At two decimal place, the effect of the altitude on the freezing point of water may appear to be absent. However, at four decimal place, the effect of the altitude become visible. Scientists and researcher must be precise in their measurement.
For instance, researcher on the Tibetan Plateau must account for the manual pressure reading of the air, since changes in air pressure can be similar to changes in the solute within the water. Furthermore, laboratory technician must be precise in their testing of water sample, as even pure water may contain mineral that affect the freezing point of the water. There are other environmental factor related to water freezing beyond those mentioned.
For instance, the gradient in which the water is exposed to the air, the microclimate in specific location, and the shape of the container in which the water is contained can affect the freezing point of water. Wind can cause water to freeze fast, but does not impact the freezing point of the water. However, impurity in the water can also lead to the water freezing at a fast rate due to the process of nucleation.
Overall, both the altitude and the solute contained within the water impact the freezing point of that water.